WHAT WAS DONE?
In a recent study [1] Peng et al. collected single-particle cryo-EM data of rabbit muscle aldolase, a somewhat challenging specimen because it’s a 149 kDa tetramer with D₂ symmetry.
Using the Apollo detector, they processed ~722,778 aldolase particles in cryoSPARC, obtaining a 3D reconstruction at 2.24 Å resolution.
The resulting density map (Figure 1) from Apollo shows very high-quality characteristics: side‐chain densities are clear, and even small features such as holes in the middle of six-membered rings are visible. These are indicators of fine atomic detail.
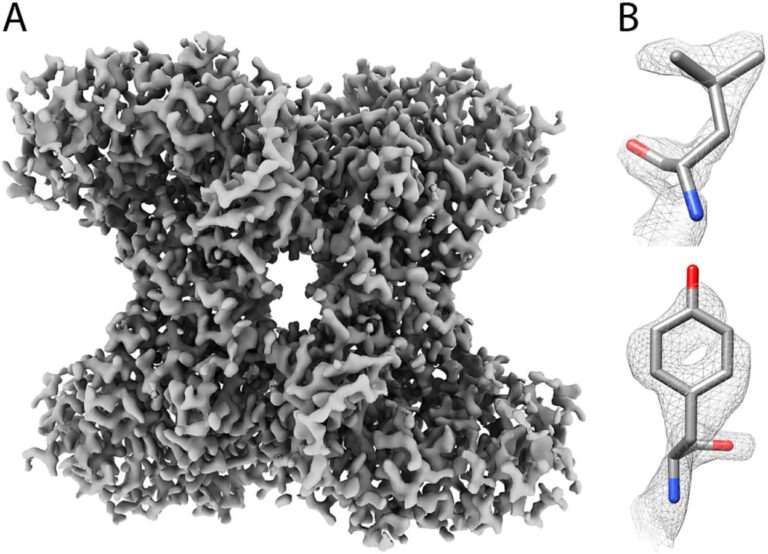
Figure 1: (A) The 2.24 Å resolution Aldolase reconstruction generated from Apollo data. (B) Examples of the sides chains showing high-resolution detail.
COMPARISONS WITH DE-64 AND K3 DETECTORS
For context, the same sample was also imaged with other detectors: the DE-64 and the K3. The reconstructions using those detectors were at lower resolution: ~2.94 Å for DE-64 with 60,000 particles, ~2.90 Å for K3 with 110,000 particles . The authors plot ResLog plots (essentially resolution vs log number of particles) , shown in Figure 2, for data collected on Apollo (red), DE-64 (green), and K3 (blue). These plots show how resolution improves as more particles are used; steeper curves or better performance per particle indicate a better detector.
Comparing the plots at 50,000 particles each is instructive. The K3 is at 3.4 Å, the DE-64 is at 3.2 Å, and Apollo is at 2.7 Å
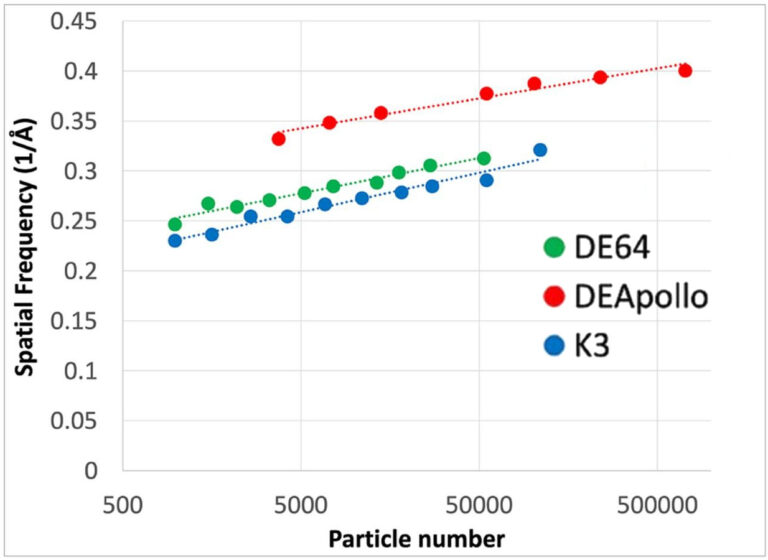
Figure 2: ResLog plot comparing the resolution of Aldolase reconstructions generated by the K3, DE-64 and Apollo direct electron detectors.
The ResLog plots show Apollo delivers sharper results per particle, outperforming both DE-64 and K3. At 50,000 particles, Apollo resolved aldolase at 2.7 Å versus 3.2 Å for DE-64 and 3.4 Å for K3.
HOW DO THE RESULTS IMPLY BETTER PERFORMANCE FROM APOLLO?
ARE THERE ANY CAVEATS?
The grids used for Apollo versus those for K3/DE-64 were not always exactly the same, although they were from the same sample preparation session. Also, in the lower-resolution data sets (K3, DE-64), many fewer particles were used (~60,000 for DE-64; ~110,000 for K3) than for Apollo. So some of the gain comes from having more data; but the curves show that even adjusting for particle numbers, Apollo’s data are superior.
CONCLUSION
The Apollo detector significantly improves the quality of cryo-EM maps compared to the K3: it delivers higher resolution structure (2.24 Å vs ~2.9 Å) and shows clearer atomic detail (side chains, ring holes). It achieves more detailed structure from comparable or even fewer data, thanks to better detector performance. For researchers wanting sharper molecular pictures, especially of smaller or less symmetric proteins, Apollo is a more powerful tool.
References:
Peng, R. et al. Characterizing the resolution and throughput of the Apollo direct electron detector. Journal of Structural Biology: X 7, 100080 (2023).