Researchers at the NIH and Yale have identified the entry receptor for HKU5, a bat merbecovirus related to MERS-CoV, using cryo-EM data collected with the Direct Electron Apollo camera.
High-resolution cryo-EM is revealing how viruses engage host receptors at the atomic level— insights that are critical for understanding how pathogens adapt and cross species barriers.
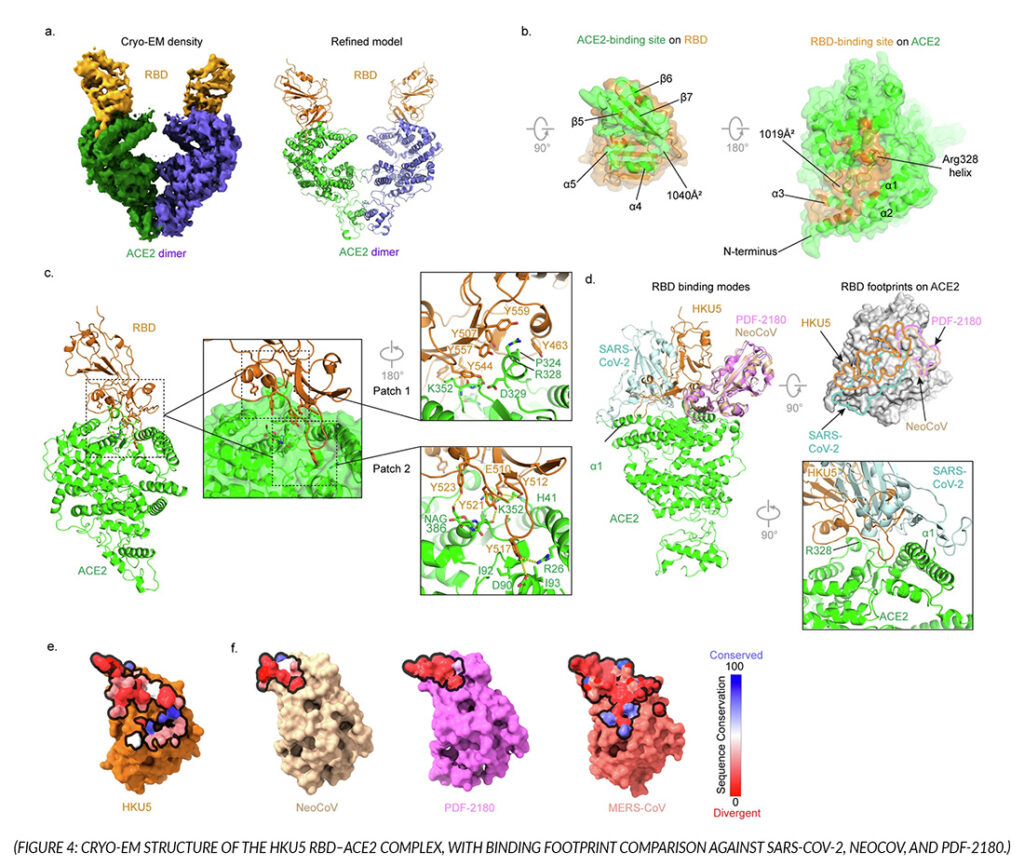
HKU5 has long been difficult to study because its cell entry receptor was unknown — a critical gap for assessing spillover risk or developing countermeasures. Alfajaro, Keeler, Li et al. screened HKU5 against a broad panel of 48 ACE2 and 31 DPP4 orthologs from wild and domestic animals. Unlike its relative MERS-CoV, which uses DPP4, HKU5 was found to use ACE2 — specifically from Pipistrellus abramus bat, American mink, and stoat.
To characterize this interaction structurally, the team collected cryo-EM data on an Apollo-equipped Titan Krios, resolving the HKU5 receptor-binding domain in complex with P. abramus ACE2 at 4.2 Å. The structure revealed a binding mode distinct from all other known ACE2-using coronaviruses, driven by a tyrosine-rich interface that targets a different region of ACE2 than SARS-CoV-2. This was confirmed through mutagenesis of both the RBD and ACE2 binding interface.
The findings have practical implications. Testing showed that neither MERS-CoV vaccine sera nor MERS-targeting monoclonal antibodies effectively neutralized HKU5, underscoring the need for broader pan-merbecovirus vaccine strategies. Notably, P. abramus bats are common in human-altered environments across East Asia, and mink are widely farmed across Asia and Europe — and during preparation of this manuscript, HKU5 was detected in diseased mink on a Chinese fur farm, consistent with the study’s predictions.
These findings highlight the value of structural virology in understanding how viruses adapt across species, and the role high-resolution cryo-EM tools play in that work.
Alfajaro, M.M., Keeler, E.L., Li, N. et al. HKU5 bat merbecoviruses engage bat and mink ACE2 as entry receptors. Nature Communications 16, 6822 (2025). Read the abstract here: https://doi.org/10.1038/s41467-025-61583-7